The Model
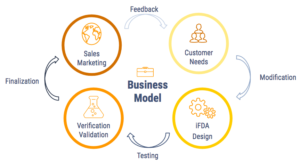
BioDrop aims for the better world, therefore, we want to develop the iFDA devices to improve the limitations of the current technologies and further serve our customer needs. We provide two types of services:
- Customization/ performance: the consultant team is available to work directly with customers to customize the devices based on their needs. We also offers testing services in which customers send in their samples and we conduct experiments. Then, they will get consulted on the results.
- Self Service: The customers can operate the system without assistance from us. They can contact us via our websites, and we will answer within 24 hours.
REGULATORY STRATEGY

The inverted floating droplet array (iFDA) is considered as Degree I because the device is made of PDMF, which is not harmful to human body. After the device is registered with FDA, we prepare the Premarket Notification (510(k)). Premarket approval (PMA) is not required for our devices since iFDA is classified as Class I. While the application is under review, we conduct various tests and publish the results to indicate the feasibility of the devices. With the FDA approval, the inverted floating droplet array is ready for market entry.
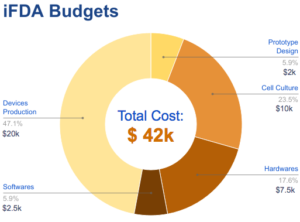
Funding requirements